Ancient Fish Used Their Lungs to Hear Underwater
Sound moves differently through water than through air. It travels faster, farther, and with more force, and the problem for a fish is that its body, being mostly water itself, offers the waves nothing to push against. They pass straight through. For a fish to actually hear, rather than simply be buffeted by pressure, it needs something inside itself that the sound can interact with: a pocket of gas, a bubble, something with density unlike the surrounding tissue. The ear alone is not enough.
This is why the discovery now being reported from fossils in eastern France matters so much. Scientists at the Natural History Museum of Geneva and the University of Geneva have found evidence that coelacanths, a lineage of lobe-finned fish with a living record stretching back more than 400 million years, once used their ossified lung to hear. The lung, long studied as a breathing organ, was apparently pulling double duty as the fish’s primary sound-detection system.
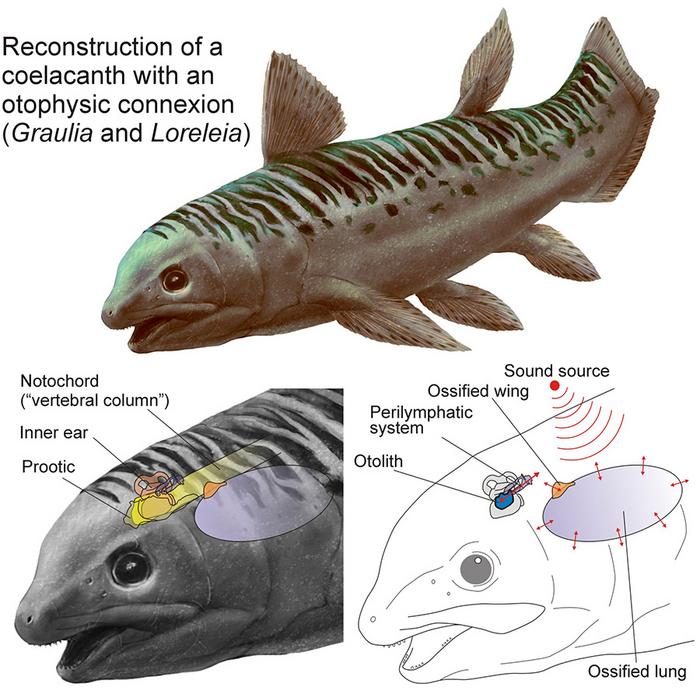
The fossils in question are two species of Triassic coelacanth, Graulia branchiodonta and the newly named Loreleia eucingulata, both roughly 240 million years old and both discovered in Lorraine in northeastern France. To study them, Luigi Manuelli, a doctoral student at the Natural History Museum of Geneva and the paper’s first author, and his colleagues used the European Synchrotron Radiation Facility in Grenoble. The particle accelerator generates X-rays powerful enough to resolve internal bone structure at the scale of a few micrometers, and it produced something the team had not fully anticipated: a pair of wing-like bony projections at the front of each animal’s ossified lung, attached where the lung met the notochord, the stiffened rod running along the spine.
The ossified lung contained a pocket of gas, and gas has a different acoustic density from the surrounding tissue and water, giving sound pressure something to push against. Bony projections on the lung, called chamber wings, were attached near the spine, and from there vibrations could travel through the notochord and into the fluid-filled canals of the inner ear. It is the same basic principle used by carp and catfish today, though they use a separate chain of small bones called the Weberian apparatus rather than the lung itself.
Living coelacanths, the genus Latimeria, adapted to deep marine environments over tens of millions of years, and their lung shrank to a vestigial structure. Without a substantial gas-filled organ to receive sound pressure, the whole system lost its function. The inner ear canals associated with hearing are still present in Latimeria but filled with connective tissue rather than fluid, a relic of a capability the animal no longer needs.
Coelacanths are lobe-finned fish, more closely related to land vertebrates than to most other fish. Finding auditory sensory cells in coelacanths that are similar to those in amphibians suggests that pressure-sensitive hearing may have developed before vertebrates left the water, and before the tympanic middle ear evolved. If confirmed in earlier fossil species, it would push the origin of vertebrate hearing significantly further back than current understanding places it.
Not entirely. Lungfish are already known to detect sound pressure through their lungs, without any specialized mechanical connection between lung and ear. Frogs use a related lung-to-ear system in their larval stage. The coelacanth case is distinctive because the lung was heavily ossified, the anatomical connection to the inner ear is reconstructable from fossils, and the lineage sits at a critical junction in vertebrate evolution, making it relevant to questions about how all backboned animals, including us, came to hear.
Those structures are called chamber wings in the new paper. Their position is what caught the researchers’ attention. They sit near the skull, close to a fluid-filled canal that runs through the cartilage of the inner ear. In the living coelacanth Latimeria chalumnae (one of only two surviving species, found today in the deep ocean off the Comoros Islands and off Sulawesi), the same canal system still exists, albeit in vestigial form, connecting the region of the inner ear to the exterior of the skull through a small opening. In the Triassic species, the team thinks this canal was the pathway: sound vibrations received by the gas-filled lung would have passed through the chamber wings into the notochord or surrounding soft tissue, and from there into the inner ear.
The analogy Manuelli draws is to a group of familiar freshwater fish. “Our hypothesis is based on analogies with modern freshwater fish such as carp or catfish,” he explains. “This system allows them to detect underwater waves and therefore hear underwater. The air bubble contained in the swim bladder is essential for detecting these waves, which would otherwise pass through the fish’s body undetected.” In carp and catfish, a chain of small bones known as the Weberian apparatus connects the swim bladder to the inner ear, doing precisely what the coelacanth’s chamber wings and canal system may have done via a different anatomical route. The gas-filled chamber is the key component in both cases. It provides the acoustic impedance mismatch, the thing that sound can push against.
What makes the case more complicated is that the lung was already understood to serve a respiratory function. The same organ, covered in bony plates arranged like overlapping roof tiles, has long been interpreted as an air-breathing adaptation. The new paper does not contest that reading; it proposes instead that the organ was doing two things at once, which is perhaps not so surprising given that lungs and swim bladders share evolutionary origins in the first place.
Inside the inner ear of L. chalumnae, Manuelli and his colleagues also found something that had not been fully mapped before: two sensory patches, a basilar papilla and what they are calling an amphibian papilla, both connected to the perilymphatic space, the fluid-filled system that in coelacanths corresponds to what carries sound pressure to auditory receptors in other vertebrates. In amphibians, these two papillae are the main sound-processing structures. Finding them in coelacanths, a lineage that diverged from the amphibian branch of the vertebrate tree long before the first animals walked on land, places their origin earlier in evolution than had been confirmed before.
The loss of the system in modern Latimeria, Lionel Cavin believes, reflects where the animals ended up. “This auditory ability was likely gradually lost as the ancestors of modern coelacanths adapted to deep marine environments,” says Cavin, curator at the Natural History Museum of Geneva and the paper’s senior author. “Their lung regressed, making this system unnecessary.” In the deep ocean, the selective pressures on hearing would have shifted; what you need to detect, and how finely you need to detect it, changes when you spend your life below 100 meters. The canal communicans, the main channel of the perilymphatic system, is still present in L. chalumnae but filled with connective tissue rather than fluid, a relic of a function the lineage no longer needs.
The question this raises, and it is one the paper handles carefully, is whether this arrangement might have been ancestral not just to coelacanths but to a broader group. Coelacanths are sarcopterygians, the lobe-finned clade that also includes lungfishes and, eventually, the land vertebrates. If the auditory papillae and the perilymphatic system that serves them were already present in a Devonian coelacanth (the team found the same canal organization in Diplocercides kayseri, a 380-million-year-old species), then some version of pressure-sensitive hearing may have predated the evolution of the tympanic membrane entirely. Our own middle ear, with its eardrum and chain of ossicles, is usually understood as the innovation that let terrestrial vertebrates hear airborne sound. But the sensory cells that receive those sound signals, the auditory papillae, may have been present and functional, detecting sound via a gas-filled internal chamber, long before any vertebrate left the water.
There are limits to what fossils can confirm here. The soft tissues that would have actually conducted vibration from the lung to the skull are gone; the team is working from bone geometry and analogy with living species. Still, the functional logic of a gas-filled chamber adjacent to a fluid-filled auditory canal is well established across multiple lineages. The coelacanth version would have been convergent with the otophysan one, arriving at a similar solution through different anatomy.
Cavin noted that the vestigial canal structures in the living coelacanth now take on new interpretive weight. The remnants in L. chalumnae, stripped of their original function but still present in the skull, are a kind of molecular palimpsest of the animal’s deeper history, and possibly of ours. How far back that history extends, whether the same pressure-detection logic was operating in the common ancestor of all sarcopterygians, is what the team is now trying to work out, probably from more Devonian material and from broader comparisons with lungfish anatomy. The answer, if it comes, will push the origin of vertebrate hearing considerably further into the past than most textbooks currently place it.
Related
Discover more from Wild Science
Subscribe to get the latest posts sent to your email.



